
Topic 20 - Organic chemistry (HL)
Question 1
HLPaper 2The reactivity of organic compounds depends on the nature and positions of their functionalgroups.
The structural formulas of two organic compounds are shown below.

Deduce, giving a reason, which of the two compounds can show optical activity.
Draw three-dimensional representations of the two enantiomers.
State the reagents used in the nitration of benzene.
State an equation for the formation of NO2+.
Explain the mechanism of the reaction between 2-bromo-2-methylpropane, (CH3)3CBr,and aqueous sodium hydroxide, NaOH (aq), using curly arrows to represent themovement of electron pairs.
Question 2
HLPaper 1Which is correct for the conversion of propanal to propyl methanoate?


Question 3
HLPaper 1What is the product of the reaction of benzene with a mixture of concentrated nitric and sulfuric acids?

Question 4
HLPaper 2Compound A is in equilibrium with compound B.

Predict the electron domain and molecular geometries around the oxygen atom of molecule A using VSEPR

State the type of hybridization shown by the central carbon atom in molecule B.
State the number of sigma (σ) and pi (π) bonds around the central carbon atom in molecule B.

The IR spectrum of one of the compounds is shown:

COBLENTZ SOCIETY. Collection © 2018 copyright by the U.S. Secretary of Commerce on behalf of the United States of America. All rights reserved.
Deduce, giving a reason, the compound producing this spectrum.
Compound A and B are isomers. Draw two other structural isomers with the formula C3H6O.
The equilibrium constant, Kc, for the conversion of A to B is 1.0×108 in water at 298 K.
Deduce, giving a reason, which compound, A or B, is present in greater concentration when equilibrium is reached.
f(i).
Calculate the standard Gibbs free energy change, ∆G⦵, in kJ mol–1, for the reaction (A to B) at 298 K. Use sections 1 and 2 of the data booklet.
f(ii).
Propanone can be synthesized in two steps from propene.Suggest the synthetic route including all the necessary reactants and steps.
g(i).
Propanone can be synthesized in two steps from propene.
Suggest why propanal is a minor product obtained from the synthetic route in (g)(i).
g(ii).
Question 5
HLPaper 1What are the E/Z designations of these stereoisomers?


Question 6
HLPaper 1Which statement is not correct regarding benzene?
Question 7
HLPaper 1Which is the correct combination of substitution reaction mechanisms?
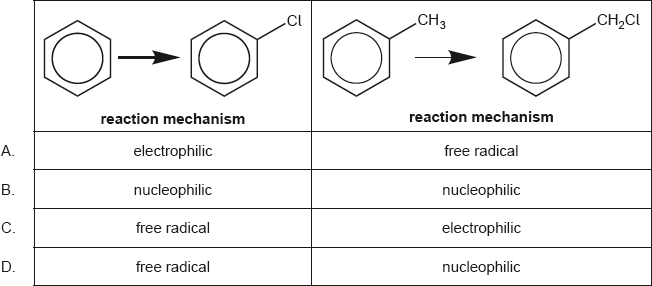
Question 8
HLPaper 1Which is a major product of the electrophilic addition of hydrogen chloride to propene?
Question 9
HLPaper 1In which compound is the halogen substituted the most rapidly by aqueous hydroxide ions?
Question 10
HLPaper 2Xylene is a derivative of benzene. One isomer is 1,4-dimethylbenzene.

Xylene, like benzene, can be nitrated.
Bromine reacts with alkanes.
State the number of 1H NMR signals for this isomer of xylene and the ratio in which they appear.
Draw the structure of one other isomer of xylene which retains the benzene ring.
Write the equation for the production of the active nitrating agent from concentrated sulfuric and nitric acids.
c(i).
Explain the mechanism for the nitration of benzene, using curly arrows to indicate the movement of electron pairs.
c(ii).
Identify the initiation step of the reaction and its conditions.
d(i).
1,4-dimethylbenzene reacts as a substituted alkane. Draw the structures of the two products of the overall reaction when one molecule of bromine reacts with one molecule of 1,4-dimethylbenzene.
d(ii).
The organic product is not optically active. Discuss whether or not the organic product is a racemic mixture.
