All topics


A pure sample of iodine-131 decays into xenon with a half-life of 8 days.
What is after 24 days?

Some transitions between the energy states of a particular atom are shown.
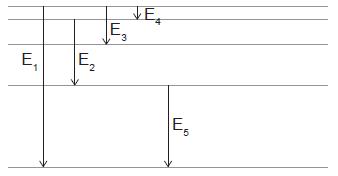
Energy transition gives rise to a photon of green light. Which transition will give rise to a photon of longer wavelength?

Which one of the following is a correct definition of the decay constant of a radio-isotope?

A radioactive sample has activity at . What will be the activity of the sample after two half-lives?

The age of the Earth is about years.
What area of physics provides experimental evidence for this conclusion?

Nucleus P decays by a sequence of emissions to form nucleus Q . One particle and two particles are emitted during the sequence. Which statement is correct?

A pure sample of an unknown radioactive nuclide has a very long half-life. For the sample the following quantities are known
I. the number of nuclei in the sample
II. the mass of the sample
III. the activity of the sample.
Which of the following will enable the half-life of the nuclide to be determined?

Radioactive decay measurement A medical physicist wishes to investigate the decay of a radioactive isotope and determine its decay constant and half-life. A Geiger-Muller counter is used to detect radiation from a sample of the isotope, as shown. 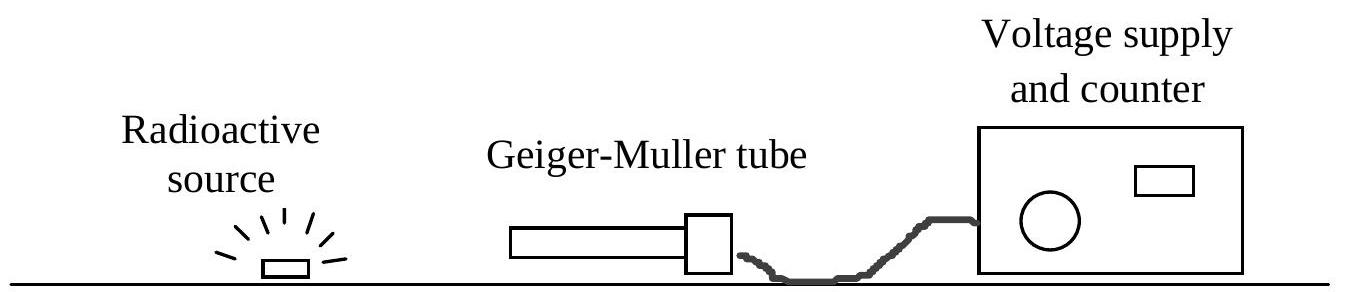 Theory predicts that the activity A of the isotope in the sample should decrease exponentially with time t according to the equation A=A₀e^(-λt), where A₀ is the activity at t=0 and λ is the decay constant for the isotope.
Theory predicts that the activity A of the isotope in the sample should decrease exponentially with time t according to the equation A=A₀e^(-λt), where A₀ is the activity at t=0 and λ is the decay constant for the isotope.

Define the activity of a radioactive sample.

Manipulate this equation into a form which will give a straight line if a semi-log graph is plotted with appropriate variables on the axes. State what variables should be plotted.

Does the plot show that the experimental data are consistent with an exponential law? Explain.

The Geiger-counter does not measure the total activity A of the sample, but rather the count-rate R of those particles that enter the Geiger tube. Explain why this will not matter in determining the decay constant of the sample.

From the graph, determine a value for the decay constant λ.

Define the half-life of a radioactive substance.

Derive a relationship between the decay constant λ and the half-life τ.

Hence calculate the half-life of this radioactive isotope.

One possible fission reaction of uranium-235 (U-235) is ^235_92U + ^1_0n → ^140_54Xe + ^94_38Sr + 2^1_0n The following data are available. Mass of one atom of U-235 = 235 u Binding energy per nucleon for U-235 = 7.59 MeV Binding energy per nucleon for Xe-140 = 8.29 MeV Binding energy per nucleon for Sr-94 = 8.59 MeV

State what is meant by binding energy of a nucleus.

Outline why quantities such as atomic mass and nuclear binding energy are often expressed in non-SI units.

Show that the energy released in the reaction is about 180 MeV.

A nuclear power station uses U-235 as fuel. Assume that every fission reaction of U-235 gives rise to 180 MeV of energy.

Estimate, in J kg^(-1), the specific energy of U-235.

The power station has a useful power output of 1.2 GW and an efficiency of 36%. Determine the mass of U-235 that undergoes fission in one day.

The specific energy of fossil fuel is typically 30 MJ kg^(-1). Suggest, with reference to your answer to (i), one advantage of U-235 compared with fossil fuels in a power station.

A sample of waste produced by the reactor contains 1.0 kg of strontium-94 (Sr-94). Sr-94 is radioactive and undergoes beta-minus (β^-) decay into a daughter nuclide X. The reaction for this decay is
^94_38Sr → X + ν_e + e^-

Write down the proton number of nuclide X.

The graph shows the variation with time of the mass of Sr-94 remaining in the sample.


State the half-life of Sr-94.

Calculate the mass of Sr-94 remaining in the sample after 10 minutes.

The energy-level diagram for an atom that has four energy states is shown.
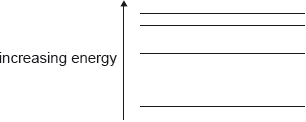
What is the number of different wavelengths in the emission spectrum of this atom?
