All topics


The diagram represents an ideal, monatomic gas that first undergoes a compression, then an increase in pressure. 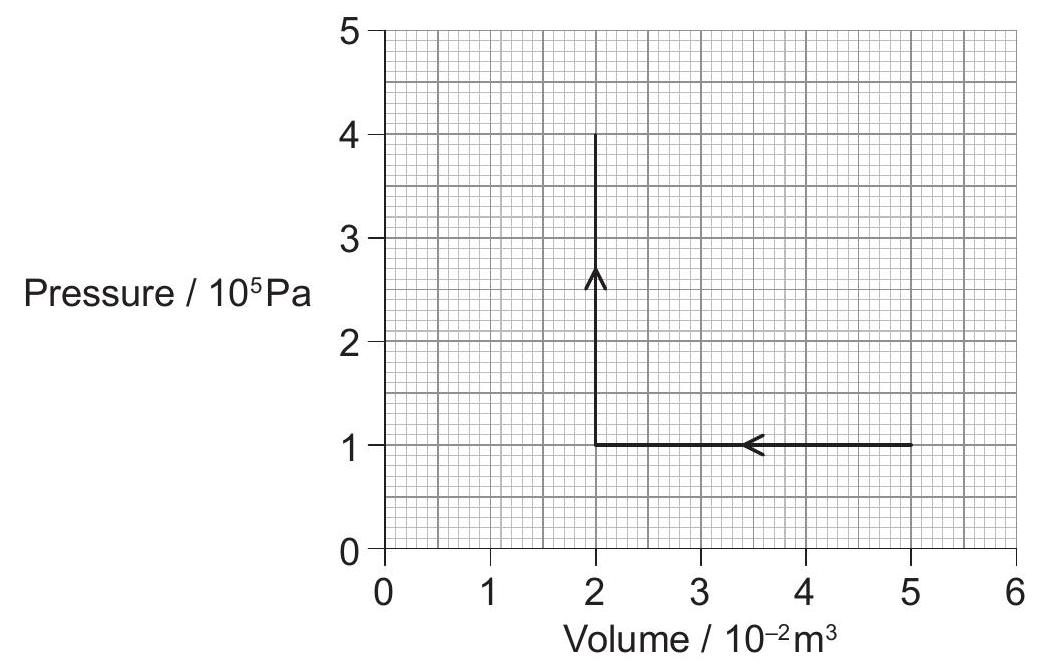

Calculate the work done during the compression.

Calculate the work done during the increase in pressure.

An adiabatic process then increases the volume of the gas to . Calculate the pressure following this process.

Outline how an approximate adiabatic change can be achieved.

It is proposed to build a heat engine that would operate between a hot reservoir at a temperature of 400 K and a cold reservoir at 300 K . See the diagram below. In each cycle it would take 100 J from the hot reservoir, lose 25 J to the cold reservoir and do 75 J of work.
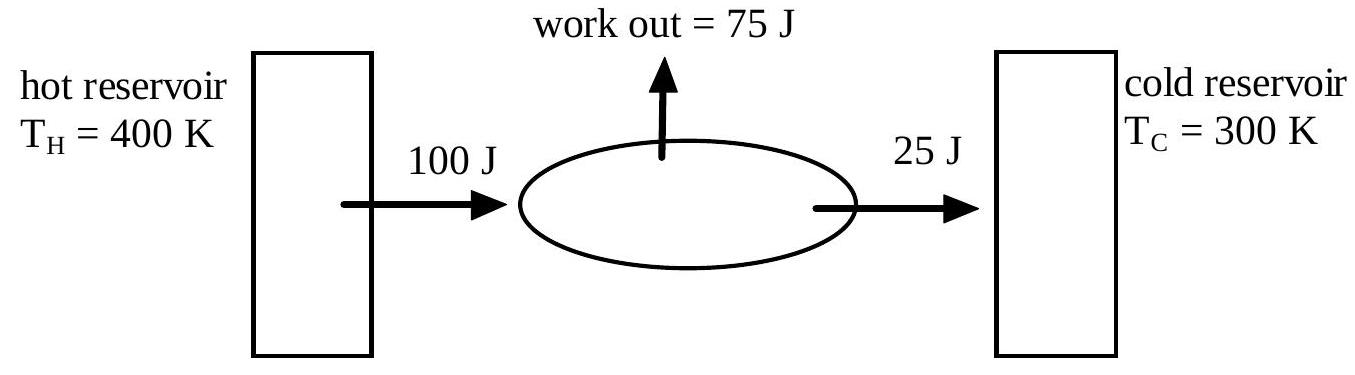
This proposed heat engine would violate

An ice cube floats in water that is contained in a tube. 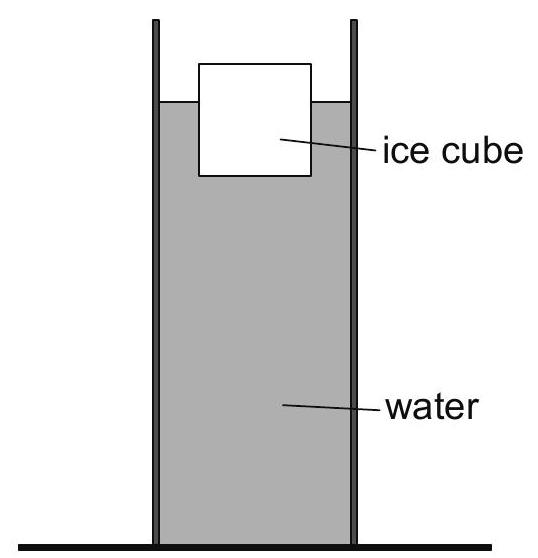 The ice cube melts.
The ice cube melts.

Suggest what happens to the level of the water in the tube.

A horizontal pipe is inserted into the cylindrical tube so that its centre is at a depth of 5.0 m from the surface of the water. The diameter D of the pipe is half that of the tube.

When the pipe is opened, water exits the pipe with speed u and the surface of the water in the tube moves downwards with speed v. Outline why u = 4v.

The density of water is 1000 kg m^-3. Calculate u.

A beaker contains water at a higher temperature than its surroundings. Which of the following correctly describes the entropy change in the water and in the surroundings as the water cools?
| entropy of the water | entropy of the surroundings |
|---|---|
| decreases | decreases |
| decreases | increases |
| increases | decreases |
| increases | increases |

The graph shows how the volume of a system varies with pressure during a cycle .
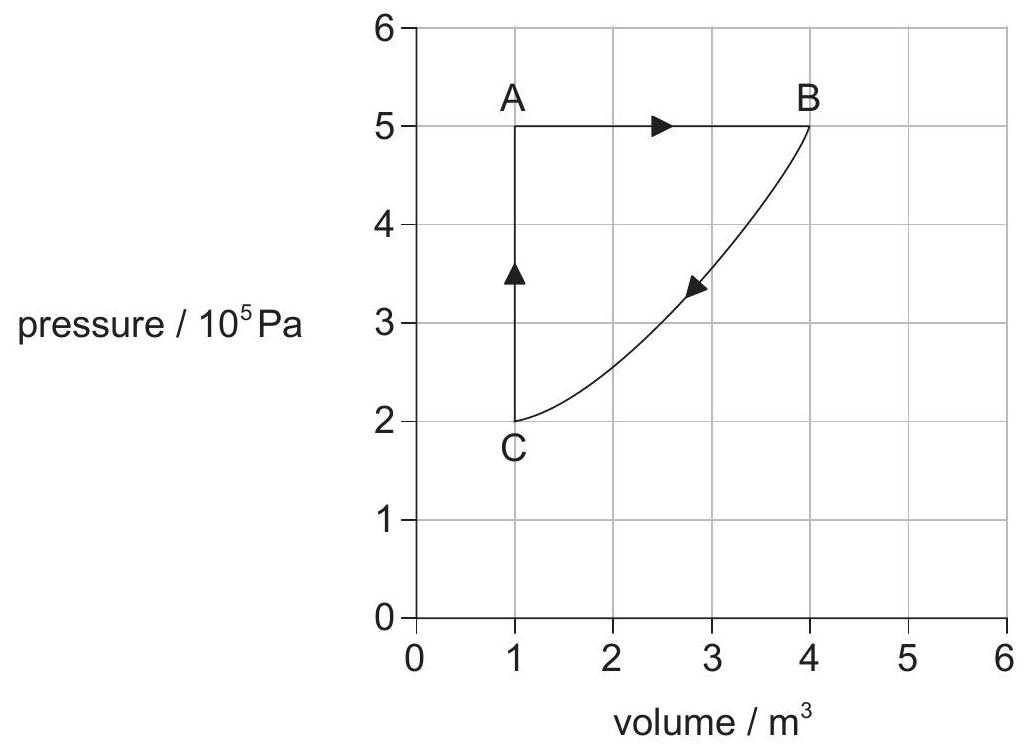
What is the work done in joules during the change ?

Which of the following is true when thermal energy is converted into work in a single process and a cyclical process?
| Single process | Cyclical process | |
|---|---|---|
| A. | complete conversion of thermal energy into work can occur | energy must be transferred from the system |
| B. | complete conversion of thermal energy into work can never occur | energy must be transferred from the system |
| C. | complete conversion of thermal energy into work can occur | energy need not be transferred from the system |
| D. | complete conversion of thermal energy into work can never occur | energy need not be transferred from the system |

A uniform rod PQ of weight is attached to a vertical wall at end P by a hinge. A force of magnitude acts on the rod at end Q as shown such that the rod is in equilibrium.
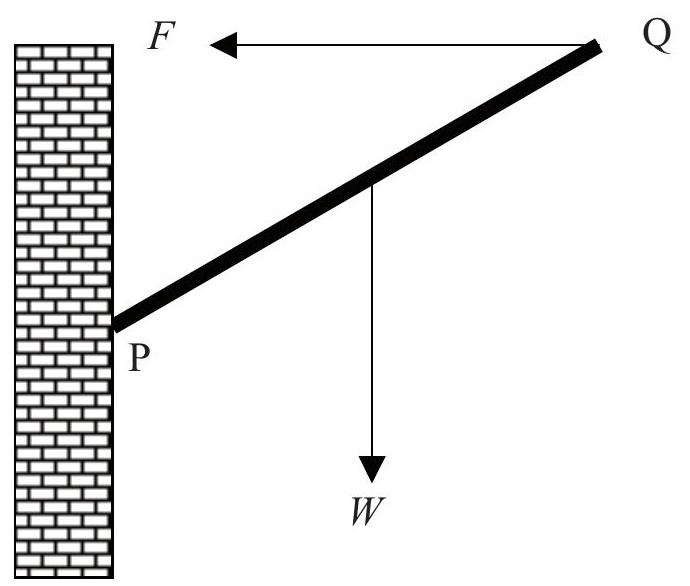
Which of the following diagrams best shows the correct direction of the reaction force at the hinge?

This question is about forces in the human body. The diagram below shows the position of a person's jawbones. 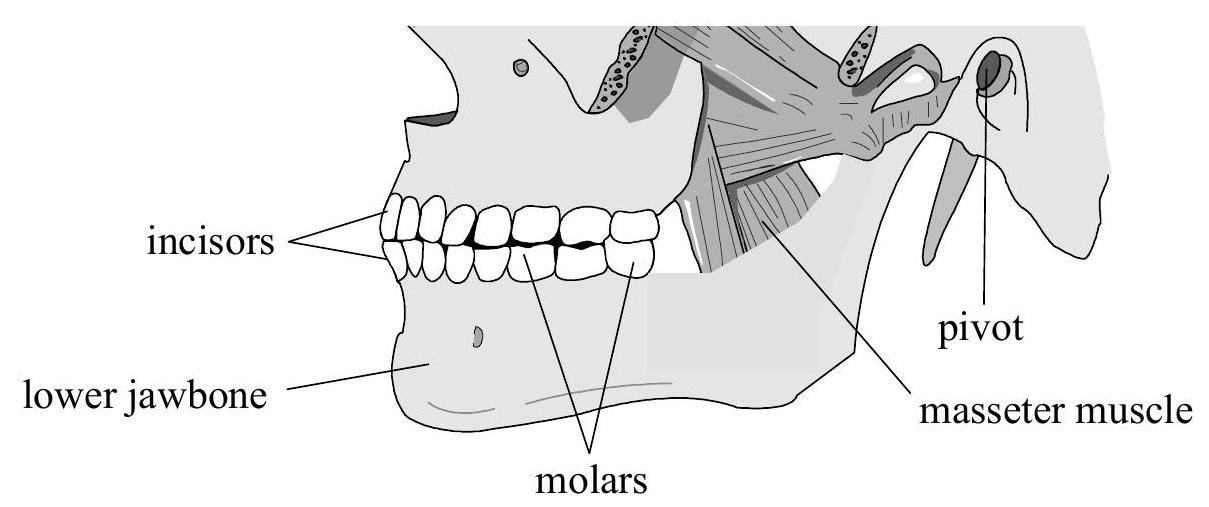 The lower jawbone may be represented by the model below.
The lower jawbone may be represented by the model below. 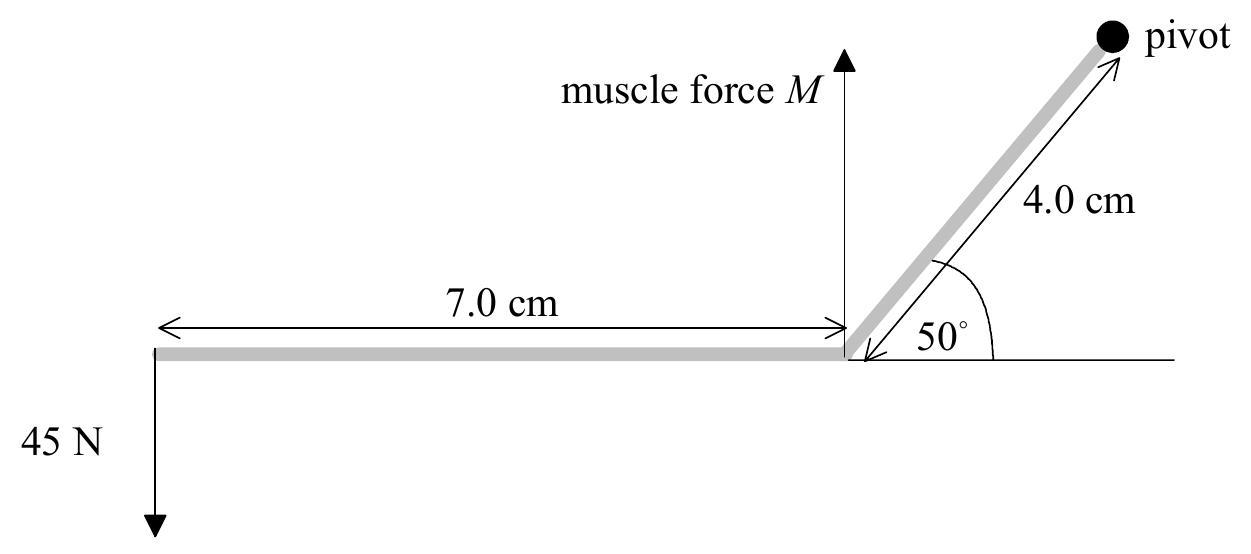 The jawbone has negligible mass. It consists of two straight parts of length 7.0 cm and 4.0 cm making an angle of 50° to each other. During one particular bite, a force of 45 N is applied by the teeth at the front of the jawbone.
The jawbone has negligible mass. It consists of two straight parts of length 7.0 cm and 4.0 cm making an angle of 50° to each other. During one particular bite, a force of 45 N is applied by the teeth at the front of the jawbone.

Calculate the magnitude M of the force applied by the masseter muscle.

Suggest why teeth used for crushing hard food are found at the back, rather than the front of the jaw.

A farmer is driving a vehicle across an uneven field in which there are undulations every 3.0 m. The farmer's seat is mounted on a spring. The system, consisting of the mass of the farmer and the spring, has a natural frequency of vibration of 1.9 Hz.

Explain why it would be uncomfortable for the farmer to drive the vehicle at a speed of 5.6 m s^-1.

Outline what change would be required to the value of Q for the mass-spring system in order for the drive to be more comfortable.

Which of the following gives the conditions for maximum amplitude in forced, but damped, oscillations?
| Driving frequency | Damping | |
|---|---|---|
| A. | greater than natural frequency | as large as possible |
| B. | equal to natural frequency | as large as possible |
| C. | greater than natural frequency | as small as possible |
| D. | equal to natural frequency | as small as possible |
